Research
Welcome to our research page!
We are interested how plants transport organic molecules to provide energy, molecular building blocks, and vital signals to distant tissues for growth and development. To this end, we study plants from the sub-cellular to the whole organism level combining microscopy, functional genetics, synthetic biology, and physiological approaches.
The plant vascular system consists of xylem and phloem, with the former transporting water and inorganic nutrients from the root to the shoot, and the phloem transporting organic nutrients, such as sugars and amino acids from photosynthetically active “source leaves” to photosynthetically inactive “sink tissues” like roots and fruits.
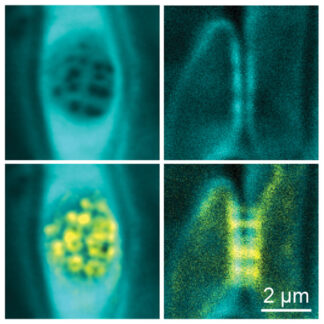
The conductive cells of the phloem, the “sieve elements” are a uniquely derived cell type. Unlike xylem vessels, sieve elements are alive. Yet they have degraded most of their cellular content and are interconnected through large cytosolic tunnels in the cell wall, the so-called “sieve pores” that perforate the “sieve plate”. At the cellular scale, sieve pores are enormous with diameters between 0.2 to 5.0 µm – approximately 10-250 times the size of plasmodesmata (the cytosolic connections between most plant cells) or 100-5000x the size of gap junctions (the cytosolic connections found between animal cells). The combination of a drastically reduced cellular content and the large connecting sieve pores make sieve elements exceptionally adapted to high-volume symplastic (i.e. intracellular) transport as opposed to the apoplastic (i.e. extracellular) transport that occurs in the dead xylem vessels. To build the large sieve pores, sieve elements undergo a profound remodeling of their cell wall that includes the deposition and degradation of callose (Barratt et al., 2011, Kalmbach & Helariutta 2019, Kalmbach et al., 2023) and pectin (Kalmbach et al., 2023). On the other hand, constrictions of sieve pores, either due to developmental defects or biotic and abiotic stressors, can have drastic physiological consequences.
In our group we investigate how these cell wall remodeling processes are orchestrated, executed, and fine-tuned during tissue development and how changes in cell wall architecture can influence phloem transport and ultimately plant growth and development. Notably, we address the localized deposition and degradation of callose and the tissue-specific remodeling of pectin.
In absolute terms, callose is a rare but usually highly localized cell wall polymer. Besides its deposition around sieve pores, callose is locally deposited in trichomes, pollen tubes, and during cytokinesis. In addition to such deposition as a context- or tissue-specific developmental feature, callose is also produced as a stress response to physical damage or pathogen infections. We aim to understand how callose biosynthetic enzymes are recruited and regulated in a tissue-specific context to shape a critical cellular feature in the developing phloem. We think that understanding callose deposition in developing sieve pores will ultimately help understanding the cellular processes of callose deposition in other developmental or adaptive contexts as well.
Unlike callose, pectin is a highly variable and, in angiosperms, the most abundant cell wall polymer. Pectin can be heavily modified and being very abundant, renders other cell wall polymers inaccessible thus potentially acting as a “gatekeeper” to enable or prevent enzymatic degradation of non-pectin cell wall polymers. Pectin-modifying enzymes are numerous and pectin breakdown products can be potent elicitors for cellular signaling pathways. This complexity makes it difficult to address pectin modification in a defined tissue- and context-specific contexts. We recently identified pectin degradation through a phloem-specific enzyme to be essential for normal sieve element development and function (Kalmbach et al., 2023). We now aim to understand how sieve-element-specific pectin modification affects phloem function and whether this pectin degradation acts solely on the cell wall architecture or rather in concert with cell wall signaling mechanisms that are locally provoked by production of pectin breakdown products.